Recently, a research team led by Dr. Liu Xianwei of the National Glycoengineering Research Center (NGRC) at Shandong University (Qingdao) published a research paper entitled “Facile Enzymatic Synthesis of Diverse Naturally-Occurring β-D-Mannopyranosides Catalyzed by Glycoside Phosphorylases” in ACS Catalysis (2020 impact factor: 12.350; 5-year impact factor: 12.741). PhD. candidate LIU Jing is the first author and Dr. Liu Xianwei is the corresponding author. The project has been consulted and assisted by Prof. WANG Peng, Prof. Gu Guofeng and Dr. Fang Junqiang. Shandong University is the first corresponding author institute.
Enzymatic glycosylation has been emerging as a promising alternative to chemical methods for the construction of varieties of glycosidic bonds. Leloir-type glycosyltransferases (GTs) which require nucleotide activated sugar donors are the primary type of enzymes capitalized as biocatalysts for the construction of glycosidic linkages. In addition to GTs, glycoside hydrolases naturally exiting to degrade glycans have also been explored for glycan assembly with or without enzyme engineering. Glycoside phosphorylases (GPases), a unique type of glycoside hydrolases, catalyze reversible reactions of glycan phosphorolysis and glycan assembly with sugar 1-phosphate as the donor substrate. Early discovered GPases are mainly involved in the degradation or assembly of glucosides. Glucoside phosphorylases have widely been explored for the production of sugar 1-phosphate and functional glucooligosaccharides. The application scope of GPases has been expanding along with the discovery of GPases with novel substrate specificities.
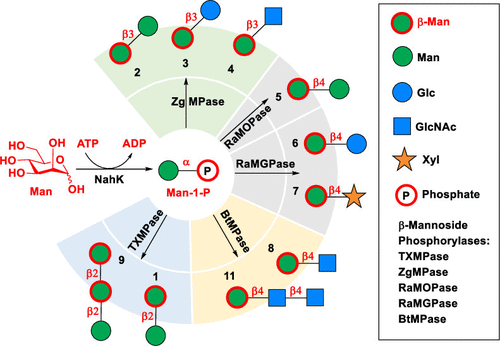
β-D-Mannopyranosides are essential components of protein N-glycans, plant β-mannans and various pathogen surface antigens. Despite their significant biological roles, the formation of 1,2-cis β-mannosyl linkage is challenging for either chemical or enzymatic synthesis. In the published article, the researchers reported a highly efficient enzymatic approach for the regioselective construction of diverse 1,2-cis β-mannosyl linkages using readily available recombinant glycoside phosphorylases. Five β-mannoside phosphorylases enabled facile synthesis of a panel of naturally-occurring β1-2-, β1-3- and β1-4-mannosides containing 8 common β-mannosyl linkages. Facile preparation of Candida antigenic β1-2-trimannoside and N-glycan core trisaccharide was achieved in satisfying yields without the need of expensive nucleotide-activated sugar donors. Mining of GPases with novel specificities and improved synthesis yields could further expand the application scope of GPase-catalyzed glycan synthesis.
This work was supported by the National Key Research and Development Program of China ,National Natural Science Foundation of China,Shandong Provincial Key Research and Development Program ,Central Government Guide Local Science and Technology Development Funds and Shandong Provincial Natural Science Foundation.
Link to article: https://doi.org/10.1021/acscatal.0c05378